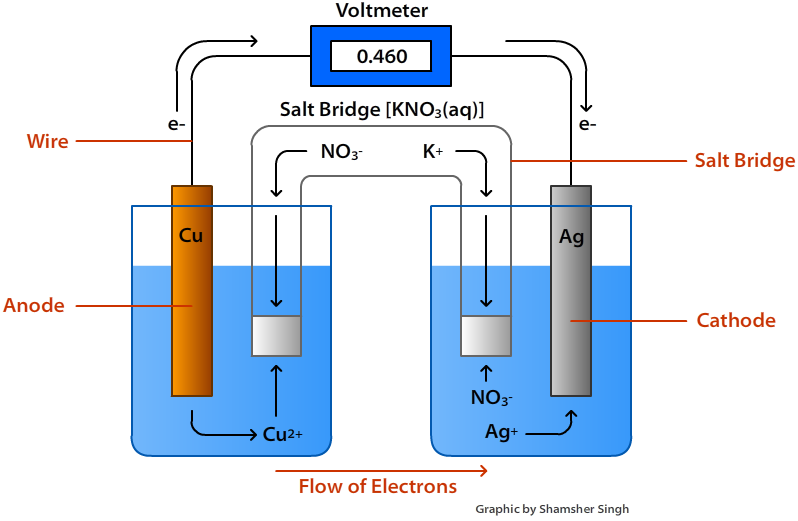
To counter that, a salt bridge is used the salt bridge contains a solution of some ionic compound whose ions migrate to either side of the voltaic cell to maintain the charge balance. Finally, because electrons are moving from one half cell to the other, a charge imbalance builds up as the reaction proceeds. Because electrons are coming from the anode, the anode is considered the negative electrode of the cell, while the cathode is considered the positive electrode of the cell. The cathode and anode collectively are the electrodes of the voltaic cell. The half cell that contains the oxidation reaction is called the anode, while the half cell that contains the reduction reaction is called the cathode. The apparatus as a whole, which allows useful electrical work to be extracted from a redox reaction, is called a voltaic (galvanic) cell.Įach individual system that contains a half reaction is called a half cell. Useful work can be extracted from the electrons as they transfer from one side to the other-for example, a light bulb can be lit, or a motor can be operated. However, in this case, the electrons transfer through the wire connecting the two half reactions that is, this setup becomes a source of electricity. One application of redox reactions requires that they be physically separated.Įven though the two half reactions are physically separated, a spontaneous redox reaction still occurs. 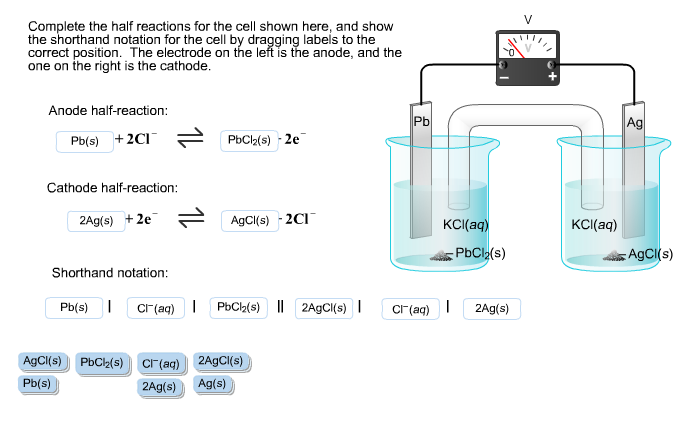 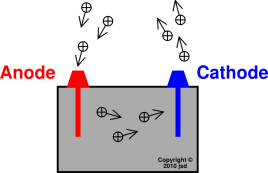
Figure 14.1 A Redox Reaction in Which the Two Half Reactions Are Physically Separated
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
